|
Consider a closed
composite system consisting of two simple systems separated by a barrier
that is impermeable to entities and pressure, but which allows the flow
of relation heat. (The physical interpretation here would be two
organizations isolated
except for the flow of memes, i.e. information. A recent major example
would be the relation between the former Soviet Union and the West.) Nk
and V are constant for each subsystem, but the energies of each
subsystem are allowed to change subject to the conservation restriction
E(1)
+ E(2) = constant
imposed by the closure
of the system as a whole. By the fundamental postulate at equilibrium
the values of E(1) and E(2) are such as to
maximize the entropy.
The condition for the
extremum is
dS = 0.
The additivity of
entropy gives
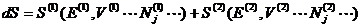
Since only E is allowed
to change,
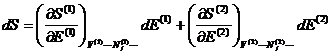
Using the definition of
temperature, this becomes
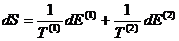
By the conservation
condition
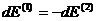
giving
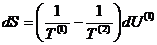
The equilibrium
condition demands that dS vanish for arbitrary values of dU(1),
therefore
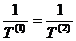
I have introduced the
quantities of heat flux and temperature without giving an interpretation
as to what they represent in a system.
In physical systems
temperature is a measure of the average kinetic energy of the
microscopic components and heat flux is a measure of the transfer of
this kinetic energy. The exact relationship between temperature and
microscopic component energies can be very complex. Examples for ideal
gases can be found in Callen [1960, Appendix D, 324-342].
In human systems
temperature is a measure of the average rate at which entities change
their relations. The mathematical demonstration of this is exactly
parallel to the demonstration of temperature and kinetic energy
relationship in physical systems. I shall not present that
demonstration here, however the result is
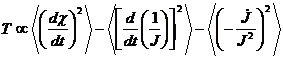 (9.4)
(9.4)
I believe it useful to
point out this relation to indicate that the concept of relation
temperature corresponds to the intuitive conception, that temperature
would be proportional to the rate of entity interdependence change. For
example, one might say that a human system with rapidly changing
relations is hot. Or, for example, a human system where the component
entities are doing the same thing the same way with the same
relationships between entities is cold, even though the entities may be
very busy. Or, for still another example, high temperature systems
contain individuals or organization of higher kinetic relation energy
and quickly invade lower temperature systems composed of lower kinetic
relation energy entities.
One can easily see that
this dynamic can be applied to the global expansion of the European
enterprise, the East India Trading Company, the geographic spreads of
the United States from East to West, and many other historical
situations. It is my intent that the application will be quantitative
not just metaphoric. However, that must wait for a bit more of the
theory development and the development of the simulation capability.
The complexity of the situation is not reducible to a set of simple
analytic expressions and must therefore be dealt with in computer based
simulations.
It is also easy to show
[Callen, 1960, 38-40] that heat flows from the higher temperature system
to the lower temperature system and that T > 0. This also corresponds
to the intuitive concept of what temperature must mean.
ç
Prior Page of Text
Next Page of Text
è
(C) 2005-2014 Wayne M. Angel.
All rights reserved. |
