|
Postulate II. There
exists a function (called the entropy S) of the extensive parameters of
any composite system, defined for all equilibrium states and having the
following property. The values assumed by the extensive parameters in
the absence of an internal constraint are those that maximize the
entropy over the manifold of constrained equilibrium states.
Callen [1960, 24]
All problems in thermodynamics are essentially
equivalent to the basic problem . . . the basic problem can be
completely solved with the aid of the extremum principle if the entropy
of the system is known as a function of the extensive parameters. The
relation that gives the entropy as a function of the extensive
parameters is known as a fundamental relation. It therefore
follows that if the fundamental relation of a
particular system is known all conceivable thermodynamic information
about the system is ascertainable there from.
The importance of the foregoing statement cannot
be overemphasized. The information contained in a fundamental relation
is all-inclusive - it is equivalent to all conceivable numerical data,
to all charts, and to all imaginable types of description of
thermodynamic properties. If the fundamental relation of a system is
known, there remains not a single thermodynamic attribute that is not
completely and precisely determined.
Postulate III. The entropy of a composite
system is additive over constituents subsystems. The entropy is
continuous and differential and is a monotonically increasing function
of the energy.
Callen [1960, 25]
Thus
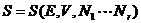 (9.1)
(9.1)
Several consequences
follow from these postulates. These consequences follow because of the
mathematical form which I postulate applies not only to physical systems
but also to human organizational systems. Since these consequences are
well described in many works, I shall simply state them here. Callen
[1960, 26], for example, gives an explanation of them.
The entropy of a
simple system is a homogenous first-order function of the extensive
parameters.
Thus if all of
the extensive parameters of a system are multiplied by a constant
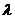 ,
the entropy is multiplied by the same constant. ,
the entropy is multiplied by the same constant.
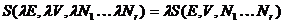 .
.
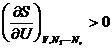 .
.
Equation (9.1) can be
inverted to yield
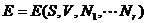 (9.2)
(9.2)
This is an alternative
form of the fundamental relation, and it also contains all thermodynamic
information about the system. In this form the entropy maximum
principle can be restated as an energy minimum principle.
At this point an
obvious question to ask is, "What is entropy? Interestingly, the theory
of thermodynamics can precede without answering this question. Since it
is not necessary and because it is well known and demonstrated in many
texts, I will not take the time to demonstrate but only state the
answer.
The
entropy of any macrostate is proportional to the logarithm of the number
of microstates associated with that macrostate.
Callen [1960, 318]
ç
Prior Page of Text
Next Page of Text
è
(C) 2005-2014 Wayne M. Angel.
All rights reserved. |
