|
In the last two
sections, I deliberately avoided the nature of the process by which a
thermodynamic engine, refrigerator, or heat pump accomplishes the
transfer of heat and work. This was done so as to emphasize that
efficiency limitations are process independent. In this section I will
consider a specific process. Continuing the analogy with physical
thermodynamics, I will discuss the Carnot cycle as it applies to human
systems.
To carry out the required process we introduce
an "auxiliary system," in addition to the two reversible heat sources
and the reversible work source. The auxiliary system is, in effect, a
tool, and at the end of the process it is left precisely in the same
state as it was in at the beginning. It is this cyclic nature of the
process within the auxiliary system that is reflective of the name of
the Carnot "cycle."
Callen [1960, 77]
The cycle is a four
step process. Changes in the temperature and entropy of the auxiliary
system are

The Carnot Cycle
Step 1: A->B. In this
step the auxiliary system is placed in thermal contact with system H, a
hot (high temperature) heat reservoir, and entity relationship contact
with system W, a reversible work source. Due to the isothermal contact
with H, the auxiliary system expands its entity relationship volume
exerting a pressure on the entity relationship space of W causing work
to be performed on W as the entity relationships are modified.
Step 2: B->C. The
thermal contact between the auxiliary system and H is removed, but the
entity relationship contact is maintained. The auxiliary system is
allowed to expand adiabatically until it temperature falls to Tc.
During this further work is done on W.
Step 3: C->D. The
auxiliary system is compressed while in thermal contact with system C, a
cold (low temperature) heat reservoir. The entity relationship contact
with W is maintained. During this step heat flows from the auxiliary
system to C, and work is transferred from W to the auxiliary system.
Step 4: D->A. The
auxiliary system is thermally isolated and compressed as it continues to
receive work from W until the temperature Th is reached. The
cycle is now complete.
The heat
withdrawn from H in process 1 is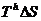 .
The heat transferred to C in process 3 is .
The heat transferred to C in process 3 is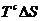 .
The difference .
The difference
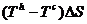 is
the net work transferred to the reversible work source in the complete
cycle. is
the net work transferred to the reversible work source in the complete
cycle.
ç
Prior Page of Text
Next Page of Text
è
(C) 2005-2014 Wayne M. Angel.
All rights reserved. |
